Question 13
NUMERICALMEDIUM
The left and right compartments of a thermally isolated container of length are separated by a thermally conducting, movable piston of area . The left and right compartments are filled with and 1 moles of an ideal gas, respectively. In the left compartment the piston is attached by a spring with spring constant and natural length . In thermodynamic equilibrium, the piston is at a distance from the left and right edges of the container as shown in the figure. Under the above conditions, if the pressure in the right compartment is , then the value of is ____
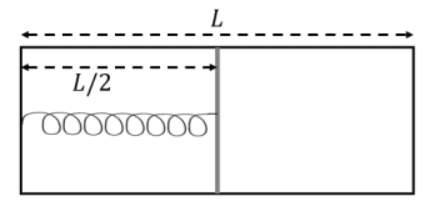
Correct Answer: 0.2
Detailed Solution
- Because the piston is thermally conducting, in equilibrium the temperatures are equal: .
- The piston is at , so the volumes are and .
- Using the ideal gas law :
- Relationship between pressures: .
- Force balance on the piston: .
- The spring is compressed/extended by . Thus .
- Substituting values into the force balance equation:
- Comparing with , we find . Final Answer: 0.2
Free Exam
Boost Your Exam Preparation!
Move beyond just reading solutions. Access our comprehensive Test Series, original Mock Tests, and interactive learning modules. Many premium tests are completely free!
- Original Mocks & Regular Test Series
- Real NTA-like Interface with Analytics
- Many Free Tests Available