Question 11
NUMERICALHARD
An ideal monatomic gas of moles is taken through a cycle consisting of consecutive adiabatic and isobaric quasi-static processes, as shown in the schematic diagram. The volume of the gas at and points are, , and , respectively. If the absolute temperature of the gas at the point is such that J ( is the universal gas constant), then the amount of heat absorbed (in J) by the gas along the path is ____
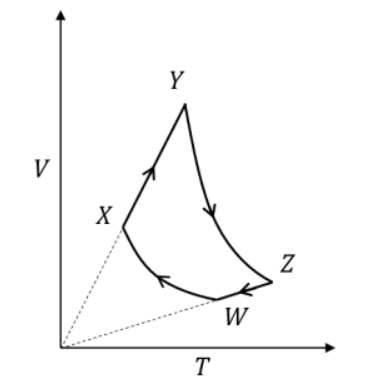
Correct Answer: 1.6
Detailed Solution
- For a monatomic gas, and .
- Path is adiabatic. For an adiabatic process, .
- Path is isobaric. For an isobaric process, heat absorbed .
- In an isobaric process, , so .
- .
- Given J, we have: Final Answer: 1.6
Free Exam
Boost Your Exam Preparation!
Move beyond just reading solutions. Access our comprehensive Test Series, original Mock Tests, and interactive learning modules. Many premium tests are completely free!
- Original Mocks & Regular Test Series
- Real NTA-like Interface with Analytics
- Many Free Tests Available