Question 7
MCQHARD
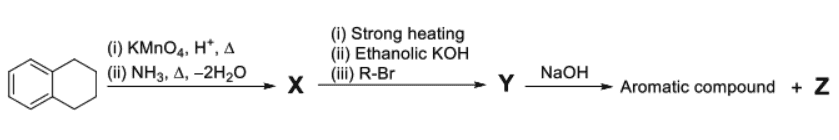
For the reaction sequence given below, the correct statement(s) is(are):
(A)
Both and are oxygen containing compounds.
(B)
on heating with forms isocyanide.
(C)
reacts with Hinsberg's reagent.
(D)
is an aromatic primary amine.
Detailed Solution
Concept: This reaction sequence is based on the oxidative cleavage of substituted benzenes, followed by the Gabriel Phthalimide Synthesis, which is a standard method for the preparation of pure primary aliphatic amines.
Step-by-step Reaction Breakdown:
- Step 1 (Formation of X): Tetralin undergoes vigorous oxidation with acidic to form Phthalic acid (benzene-1,2-dicarboxylic acid). When phthalic acid is treated with and heated (), it forms Phthalamide (). (Note: The explicit loss of indicates the formation of phthalamide).
- Step 2 (Formation of Y): * (i) Strong heating of Phthalamide () causes the loss of an molecule, resulting in ring closure to form Phthalimide.
- (ii) Reaction with ethanolic forms Potassium phthalimide (a strong nucleophile).
- (iii) Reaction with an alkyl halide () results in an substitution, yielding -alkylphthalimide ().
- Step 3 (Formation of Z): Alkaline hydrolysis (using ) of -alkylphthalimide () yields Sodium phthalate (the aromatic compound) and a primary aliphatic amine, ().
Evaluation of Options:
- (A) Both X and Y are oxygen containing compounds: True. (Phthalamide) and (-alkylphthalimide) both contain carbonyl oxygen atoms.
- (B) Y on heating with forms isocyanide: False. is an imide, not a primary amine. Only primary amines give the positive isocyanide (carbylamine) test.
- (C) Z reacts with Hinsberg's reagent: True. () is a primary amine, which reacts with Hinsberg's reagent (Benzenesulfonyl chloride, ) to form an -alkylbenzenesulfonamide that is soluble in alkali.
- (D) Z is an aromatic primary amine: False. The Gabriel phthalimide synthesis cannot be used to prepare aromatic primary amines because aryl halides do not undergo nucleophilic substitution () with the phthalimide anion under normal conditions. Thus, is strictly an aliphatic primary amine.
Final Answer: Options (A) and (C) are correct.
Free Exam
Boost Your Exam Preparation!
Move beyond just reading solutions. Access our comprehensive Test Series, original Mock Tests, and interactive learning modules. Many premium tests are completely free!
- Original Mocks & Regular Test Series
- Real NTA-like Interface with Analytics
- Many Free Tests Available