Question 4
The correct reaction/reaction sequence that would produce a dicarboxylic acid as the major product is
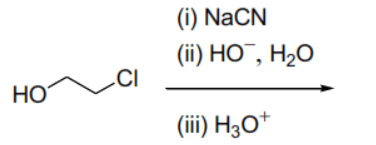
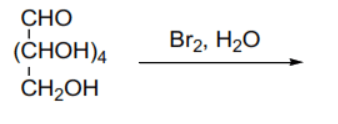

Detailed Solution
(A) Nucleophilic substitution of chloride by cyanide followed by hydrolysis results in the formation of 3-hydroxypropanoic acid (), which is a monocarboxylic acid.
(B) Oxidation of glucose with bromine water () selectively oxidizes the terminal aldehyde group to a carboxylic acid group, producing gluconic acid (), which is a monocarboxylic acid.
(C) Bromocyclohexane undergoes dehydrohalogenation with to form cyclohexene. Vigorous oxidation of cyclohexene with hot acidic () cleaves the double bond to form adipic acid (), which is a dicarboxylic acid.
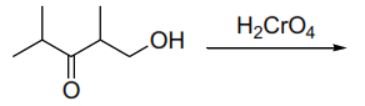
(D) Reaction of the keto-alcohol 4-hydroxy-3-methylbutan-2-one with (Jones reagent) oxidizes the primary alcohol group to a carboxylic acid group while the ketone remains unchanged, resulting in a keto-acid (monocarboxylic acid).
Therefore, reaction sequence (C) is the only one that produces a dicarboxylic acid.
Boost Your Exam Preparation!
Move beyond just reading solutions. Access our comprehensive Test Series, original Mock Tests, and interactive learning modules. Many premium tests are completely free!
- Original Mocks & Regular Test Series
- Real NTA-like Interface with Analytics
- Many Free Tests Available