Question 3
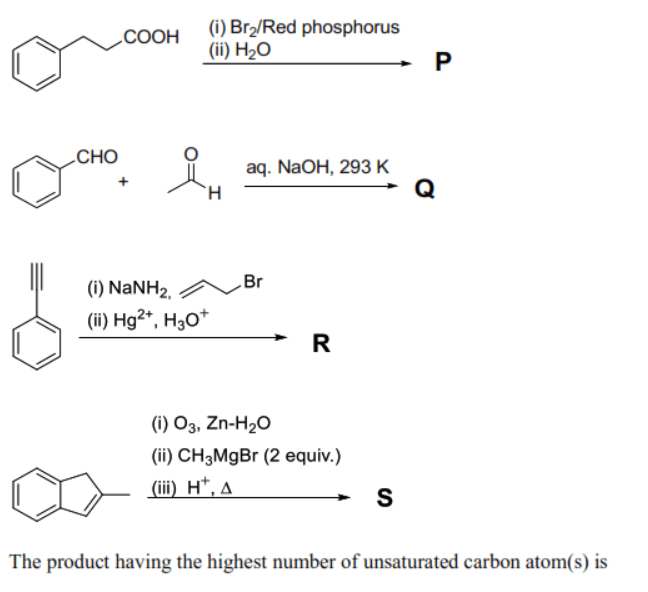
Monocyclic compounds and are the major products formed in the reaction sequences given below in the figure.
Detailed Solution
-
In reaction 1, 3-phenylpropanoic acid undergoes the Hell-Volhard-Zelinsky (HVZ) reaction to produce 2-bromo-3-phenylpropanoic acid (). The unsaturated carbons include the 6 carbons of the benzene ring and the 1 carbon of the carboxylic acid carbonyl, totaling 7.
-
In reaction 2, Benzaldehyde and Formaldehyde undergo a Cross-Cannizzaro reaction in the presence of . Formaldehyde is oxidized to sodium formate, and benzaldehyde is reduced to benzyl alcohol (). The number of unsaturated carbons in the monocyclic product (benzyl alcohol) is 6 (from the benzene ring).
-
In reaction 3, ethynylbenzene (phenylacetylene) reacts with to form an acetylide ion, which then reacts with allyl bromide to yield 1-phenylpent-4-en-1-yne. Subsequent hydration with (Markovnikov addition) yields 1-phenylpent-4-en-1-one (). The number of unsaturated carbons is 6 (benzene ring) + 1 (ketone carbonyl) + 2 (terminal alkene) = 9.
-
In reaction 4, Indene undergoes reductive ozonolysis to form 2-(2-oxoethyl)benzaldehyde. This dialdehyde/keto-aldehyde reacts with 2 equivalents of to form a diol. Subsequent acid-catalyzed dehydration () produces a product () with two external double bonds conjugated with the benzene ring (e.g., 1-(prop-1-en-1-yl)-2-vinylbenzene). The number of unsaturated carbons is 6 (benzene ring) + 2 (from first alkene side chain) + 2 (from second alkene side chain) = 10.
Comparing all products, contains the highest number of unsaturated carbon atoms (10).
Boost Your Exam Preparation!
Move beyond just reading solutions. Access our comprehensive Test Series, original Mock Tests, and interactive learning modules. Many premium tests are completely free!
- Original Mocks & Regular Test Series
- Real NTA-like Interface with Analytics
- Many Free Tests Available