Question 7
MCQMEDIUM
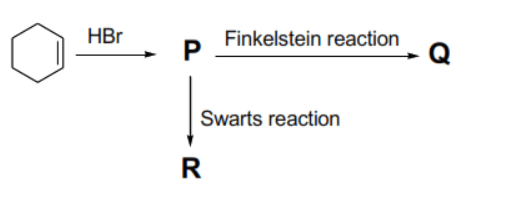
For the reaction sequence given below, the correct statement(s) is/are:
(In the options, is any atom other than carbon and hydrogen, and it is different in and )
(A)
bond length in and follows the order .
(B)
bond enthalpy in and follows the order .
(C)
Relative reactivity toward reaction in and follows the order .
(D)
value of the conjugate acids of the leaving groups in and follows the order .
Detailed Solution
First, identify the halogen atom in each compound:
- Reaction of cyclohexene with yields bromocyclohexane (). Thus, in .
- The Finkelstein reaction typically involves the exchange of a halide (like ) with (using in acetone). Thus, is iodocyclohexane, and in .
- The Swarts reaction involves the exchange of a halide with (using metallic fluorides like ). Thus, is fluorocyclohexane, and in .
Now, evaluate the statements based on the halogen properties ():
- (A) Bond Length: Atomic size follows the order . Consequently, , which means the order is . Statement (A) is incorrect.
- (B) Bond Enthalpy: Bond enthalpy is generally inversely proportional to bond length. The order of bond strength is , which means . Statement (B) is correct.
- (C) Reactivity: Reactivity depends on leaving group ability, which follows the order . Thus, the reactivity order is . Statement (C) is incorrect.
- (D) of Conjugate Acids: The conjugate acids of the leaving groups are , , and . Their values are approximately: , , and . The order of is , which translates to . Statement (D) is incorrect.
Free Exam
Boost Your Exam Preparation!
Move beyond just reading solutions. Access our comprehensive Test Series, original Mock Tests, and interactive learning modules. Many premium tests are completely free!
- Original Mocks & Regular Test Series
- Real NTA-like Interface with Analytics
- Many Free Tests Available