Question 4
Consider the depicted hydrogen () in the hydrocarbons given below. The most acidic hydrogen () is
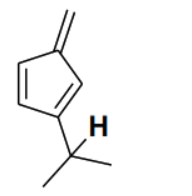
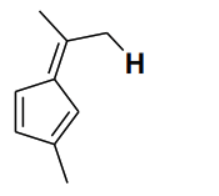
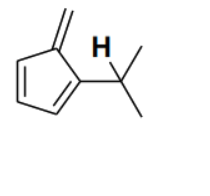
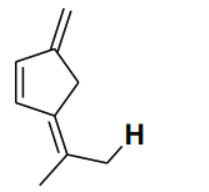
Detailed Solution
The acidity of a hydrogen atom in a hydrocarbon is determined by the stability of the conjugate base (carbanion) formed after the removal of the proton (). In option (B), the removal of the depicted hydrogen results in a carbanion where the negative charge is directly adjacent to a five-membered ring system. This negative charge can delocalize into the ring, forming a cyclopentadienyl anion. The cyclopentadienyl anion is aromatic because it is planar, cyclic, fully conjugated, and contains electrons ( where ). The stabilization gained through aromaticity makes the conjugate base extremely stable, and thus the corresponding hydrogen is the most acidic among the given options.
Boost Your Exam Preparation!
Move beyond just reading solutions. Access our comprehensive Test Series, original Mock Tests, and interactive learning modules. Many premium tests are completely free!
- Original Mocks & Regular Test Series
- Real NTA-like Interface with Analytics
- Many Free Tests Available