Question 13
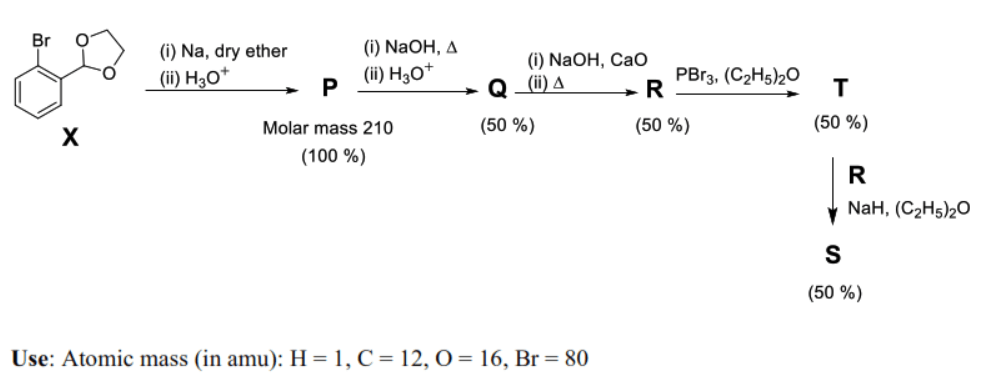
The reaction sequence given below is carried out with 16 moles of . The yield of the major product in each step is given below the product in parentheses. The amount (in grams) of produced is ________.
Detailed Solution
-
Step 1 (): 16 moles of undergo Wurtz-type coupling followed by acid hydrolysis of the acetal groups. Two moles of () react to form one mole of the dialdehyde (, ).
- Molar mass of g/mol (matches given data).
- Moles of moles.
-
Step 2 (): Intramolecular Cannizzaro reaction of gives ().
- Moles of moles.
-
Step 3 (): Soda-lime decarboxylation of removes the group to form (, ).
- Moles of produced moles.
-
Step 4 (): Conversion of alcohol to bromide (, ).
- Moles of mole.
- Note: From the 2 moles of produced in Step 3, 1 mole was consumed to make , leaving 1 mole of unreacted.
-
Step 5 (): Williamson ether synthesis between (1 mole) and the remaining (1 mole) using . is bis([1,1'-biphenyl]-2-ylmethyl) ether ().
- Moles of moles.
- Molar mass of g/mol.
-
Final Calculation:
- Amount of grams.
Boost Your Exam Preparation!
Move beyond just reading solutions. Access our comprehensive Test Series, original Mock Tests, and interactive learning modules. Many premium tests are completely free!
- Original Mocks & Regular Test Series
- Real NTA-like Interface with Analytics
- Many Free Tests Available